 | |
| Names | |
|---|---|
| IUPAC names
Molybdenum(III) iodide Molybdenum triiodide | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | MoI3 |
| Molar mass | 476.65 g/mol |
| Appearance | black solid |
| Melting point | 927 °C (1,701 °F; 1,200 K) (decomposes) |
| Solubility in water | insoluble |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Molybdenum(III) iodide is the inorganic compound with the formula MoI3.
Preparation
Molybdenum(III) iodide is created by the reaction of molybdenum hexacarbonyl with iodine gas at 105 °C (221 °F).
- 2 Mo(CO)6 + 3 I2 → 2 MoI3 + 12 CO
It can also be made from molybdenum(V) chloride and a solution of hydrogen iodide in carbon disulfide.
- MoCl5 + 5 HI → MoI3 + 5 HCl + I2
A further method is direct reaction between molybdenum metal and excess iodine at 300 °C (572 °F).
- 2 Mo + 3 I2 → 2 MoI3
As molybdenum(III) iodide is the highest stable iodide of molybdenum, this is the preferred route.
Properties
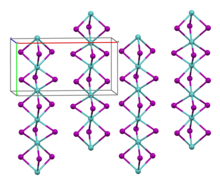
Molybdenum(III) iodide is a black antiferromagnetic solid that is air-stable at room temperature. In vacuum, it decomposes above 100 °C to molybdenum(II) iodide and iodine. It is insoluble in polar and non-polar solvents. Its crystal structure is isotypic with zirconium(III) iodide.
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1019–1021. ISBN 978-0-08-037941-8.
- ^ hrsg. von Georg Brauer. Unter Mitarb. von M. Baudler (1981). Handbuch der präparativen anorganischen Chemie / 3 (3rd ed.). Stuttgart: Enke. p. 1539. ISBN 3-432-87823-0. OCLC 310719495.
- Riedel, Erwin; Christoph, Janiak; Meyer, Hans-Jürgen (2012). Riedel moderne anorganische Chemie. Riedel, Erwin, 1930-, Janiak, Christoph., Meyer, Hans-Jürgen. (4. Aufl ed.). Berlin: De Gruyter. p. 357. ISBN 978-3-11-024900-2. OCLC 781540844.
| Molybdenum compounds | |
|---|---|
| Mo(0) | |
| Mo(II) | |
| Mo(III) | |
| Mo(IV) | |
| Mo(V) | |
| Mo(VI) | |
| Salts and covalent derivatives of the iodide ion | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||